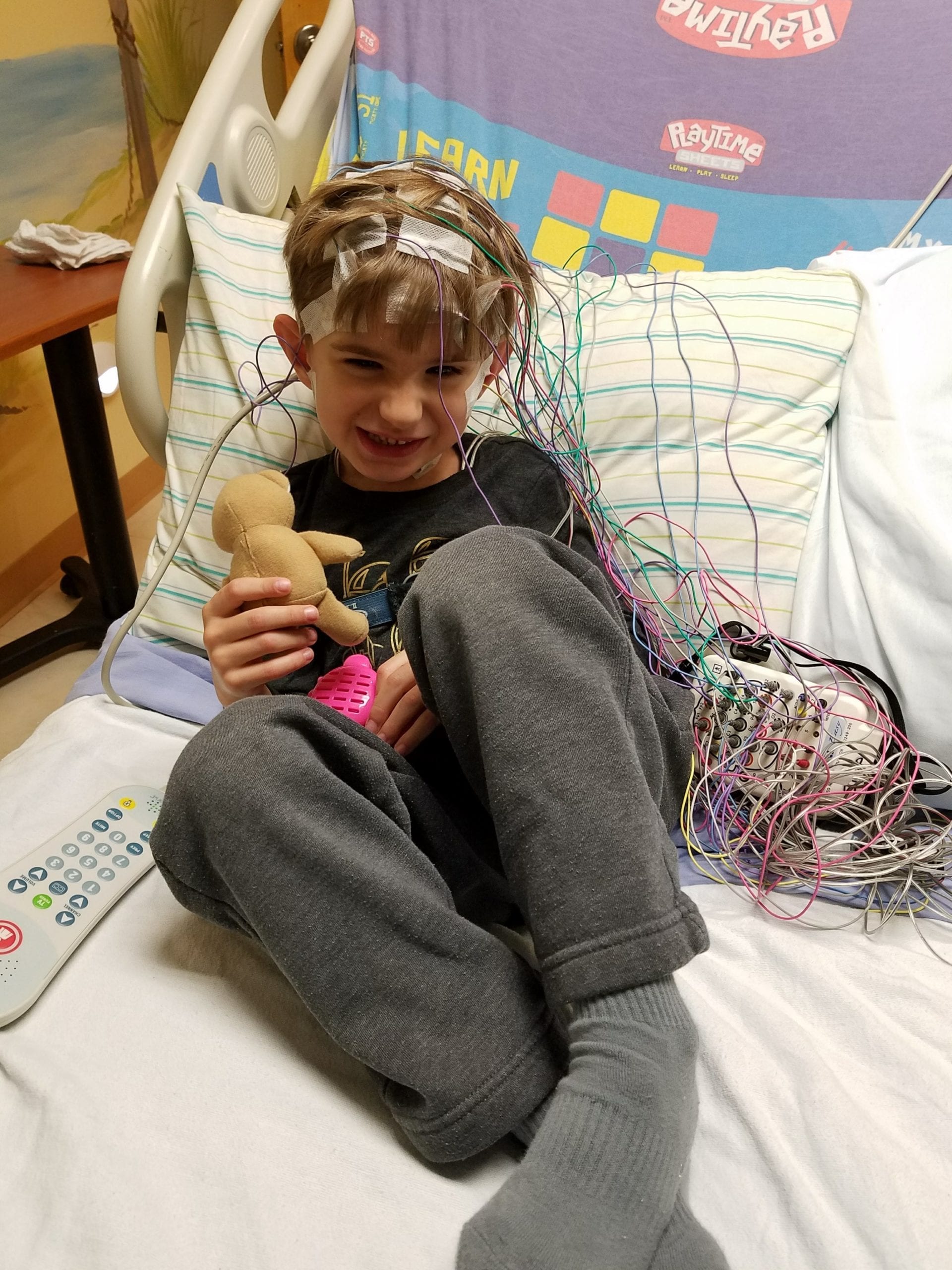
Current Research Opportunities
Are you interested in having your family or your child with chromosome 15q11.2-13.1 duplication syndrome participate in research studies? Some of the studies may involve only a phone interview, questionnaire or mailing of information. Some studies may require you to travel to other cities. Others may involve blood tests, EEG’s and other medical procedures.
As part of our mission, Dup15q Alliance seeks to unite families, researchers, and professionals; and promote research, awareness, and empower individuals with dup15q syndrome by advancing breakthrough research and life-changing therapeutic treatments. Dup15q Alliance formally endorses and funds research and collaborates with researchers interested in research on chromosome 15q duplications by disseminating research information and promoting opportunities for Dup15q Alliance families to participate in research studies and clinical trial opportunities.
The Dup15q Alliance is determined to take our family and patient involvement to the next level. We NEED to find better treatments for all of the symptoms of dup15q syndrome. We NEED to know how those with dup15q syndrome develop over time. We NEED to know why some children affected by dup15q syndrome have seizures and some don’t. We NEED to emphasize the importance of research. Direct patient and family research is critical to meet these needs, identify other needs, get better treatments and make positive changes to the lives of all those affected by dup15q syndrome. We may be rare but we are not small. We have several opportunities as a powerful community to drive our researchers to do better. The more we participate, the more they hear us and our outcry for answers.
Sign up for our NEW Research and Clinical Trial Text Alerts! You can unsubscribe from text alerts at any time.
Use this link below or Text “Alerts” to (847) 744-8904 to sign up!
Dup15q Research Registry (LADDER Database)
We are collecting data and tracking the development and care of families affected by dup15q, including diagnostics, medications, how meds are working, how other treatments are working, and getting full picture of dup15q syndrome.
Advancing Research is our best chance of finding new and improved therapeutics for dup15q syndrome.
LADDER was developed by the ASF and Dup15q Alliance, in partnership with RTI International, in response to a critical need for a centralized database to collect and manage the information provided by parents, caregivers, clinicians, and researchers about the natural history and clinical needs of patients with Angelman or Dup15q.
How Does LADDER Advance Research?
Before LADDER, information about an individual with dup15q from research studies, doctor visits and registries was stored in disparate systems. There was no connection or way to link a research participant to their information from a clinic visit.
Gathering the information in one place and connecting it, creates a higher level of understanding of dup15q which increases the potential for future discoveries that will lead to clinical trials and treatments. In addition, we will better understand how development, behavior and clinical needs change over time.
The data in LADDER can be accessed by:
- Physicians who treat dup15q syndrome
- Researchers who are working to find treatments and a cure
- Pharmaceutical partners who are working on drug development projects
How You Can Help:
Family members of individuals with dup15q syndrome are important allies in the road to developing treatments for these conditions and, in many cases, families have already contributed data elsewhere. LADDER Learning Network and LADDER Database maximizes those contributions by combining those data into a centralized platform, which streamlines research efforts focused on answering important questions about dup15q.
Register
Not sure if you already had an account from the past Dup15q International Medical Registry? Email us at 15qnetworkcoordinator@dup15q.org to check for you!
Disability Language Preferences Study
The Genetic Alliance is seeking diverse perspectives about the lived experience of how language interacts with disability and health. We aim to explore language preferences between those who are personally affected by a health condition or disability and unaffected individuals who are advocates, healthcare workers, or loved ones of affected individuals.
Everyone is invited – including those with diverse neurotypes, physical differences, mental health conditions, chronic illnesses, genetic diagnoses, and other health conditions.
Participation in this research study would involve the completion of a short survey. The survey should take 15 minutes or less to complete. Click the link below to get started.
https://id.lunadna.com/referrer/peer-group-uncg1?studyName=disability-uncg
EEG Database
Upload your EEG/MRI to the LADDER Database. Don’t have an account with LADDER? Register Here
Don’t have a copy of your child’s EEG or MRI? Here is a sample letter/email to send to your doctors office to obtain copies of research.
“Dear Dr.
,
My child is a patient of yours and as you know we are part of a large dup15q community dedicated to understanding characteristics of dup15q syndrome. In order to help understand the syndrome better, the Dup15q Alliance (patient advocacy group) is supporting a research study that can put our son/daughter’s EEG to be compared with other kids with the syndrome. Additionally, this research study also investigates overnight sleep EEGs in Dup15q children, to better inform sleep disturbances. In order for us to participate in this study, we need a copy of our child’s EEG recording. This can be a few minutes of clinical EEG recording and/or overnight sleep EEG recording, whichever was collected at your clinic during our visit. Along with EEG, any other relevant medical information collected during the visit would be great to have. It will be great if you could let us know of any paperwork involved to get this process started. We are very eager to participate in this research study and very much appreciate your help in sending us the recordings and medical information collected during our visit.
Thank you!”
HELP Represent Dup15q in Rare Epilepsy with ECHO Survey
WHO: Parents and Caregivers of Individuals with Rare Epilepsy.
WHAT: Help inform a first-of-its-kind Rare Epilepsy ECHO launching early 2023. Funded by CDC grant to educate targeted HCPs about rare epilepsies.
WHERE: Fill out a brief survey here.
WHY: Rare epilepsy diagnoses are on the rise. With thousands of individual diseases, even the most experienced practitioners are unable to be knowledgeable about every disorder (best practices for diagnosis, treatment, and management). The Rare Epilepsy ECHO seeks to disseminate rapidly changing rare epilepsy knowledge from experts to diverse health care practitioners to improve clinical care and patient outcomes.
Ciitizen Research Opportunity
Dup15q Alliance has been presented with a new opportunity provided by Invitae, a leading medical genetics company, through their health data platform, Ciitzen. This platform will rapidly provide researchers and pharmaceutical companies YEARS worth of de-identified data that will also have the ability to link back to the Dup15q LADDER Database with your consent.
What is Ciitizen?
Ciitizen is an online technology platform that helps patients get more out of their health records while providing the opportunity to advance research for disorders like dup15q syndrome.
How does Ciitizen work?
With your consent, Ciitizen will request your child’s medical records on your behalf and load them to their profile. Clinical data will be organized and summarized, free of charge. Caregivers retain full control of the patient’s health records. This gives parents years worth of records in a easy to follow format.
How does Ciitizen advance research?
With your consent, clinicians, researchers and biopharma can access deidentified data to aid in research through Ciitizen’s databank of medical and survey data collected from children and adults living with rare disorders, to be used for future research needs in collaboration with academic and pharmaceutical researchers (the “Databank”). This data can also be linked back to the Dup15q LADDER Database
Joining takes less than 10 minutes
All you have to do to join this free platform is:
- Go to ciitizen.com/dup15q
- Provide your photo ID
- Provide your child’s birth certificate or guardianship paper
- Provide a list of healthcare providers – neurologists, hospitals, specialists etc. We know this one alone may seem daunting. When you provide the providers name, and contact information, Invitae does the rest. They reach out and get years worth of records that you will later be able to login to and share with your providers or view for your child’s needs.
Need help getting signed up?
Contact justin.barreto@invitae.com.
Mobile: 317-832-3072
COVID-19 Experiences in the Rare Epilepsy/DEE community
COVID-19 Experiences in the Rare Epilepsy/DEE community
This survey is being conducted by DEE-P Connections, a collaborative project run by Wishes for Elliott.
The Dup15q Alliance is proud to be a partner of the DEE-P initiative (the Developmental and Epileptic Encephalopathies-Project), which is dedicated to serving the distinct needs of families with children most severely affected by DEEs. https://www.deepconnections.net/partner-with-us
Developmental and Epileptic Encephalopathies (DEEs) are defined as a group of rare neurodevelopmental disorders, characterized by early-onset seizures that are often intractable with a developmental delay.
Cellular Phenotypes in Dup15q Syndrome
EXPECT Survey
TAKE THE SURVEY HERE
Together with David Mandell from U. Penn, Autism Science Foundation has recently launched the EXPECT survey: Experiences of Participants Enrolled in Clinical Trials. The goal is to obtain feedback from people who both did or did not participate in an in person research study and about their experiences including why they would or would not do one again, or why they have never participated in one. It involves a 20-30 minute survey and anyone who wishes to share their email address will be entered to win an iPad. The results will be used to focus efforts on ensuring participation in research studies, including clinical trials, across idiopathic and rare genetic forms of ASD.
The Global Pediatric Epilepsy Surgery Research Project
Dup15q Alliance has partnered with the Brain Recovery Project to see if surgical interventions “work” on Dup15q Syndrome related seizures.
The Global Pediatric Epilepsy Surgery registry is a research project that allows families to share their experiences with and without epilepsy surgery in childhood by completing a series of surveys. The data collected will be used to answer some of the most important questions about pediatric epilepsy surgery, such as:
What can a child do over time after epilepsy surgery?
What post-operative therapies and interventions are useful?
What are some unusual symptoms or medical issues that happen after epilepsy surgery that parents report but doctors may be unaware of?
What are the issues that concern parents?
What is the impact of epilepsy surgery on a child’s quality of life?
What surgical techniques work best and for whom?
The main purpose of the registry is to gather information that can be used to understand the developmental trajectory after epilepsy surgery. Researchers can then use the registry to analyze the information, look for changes in a child’s function over time, determine whether some surgical procedures are more effective at stopping seizures than others, recruit participants to their approved studies, and collect new information by adding new questions to the registry.
More Info
Has having Telehealth for your family been helpful?
Congress will need to act soon on which Covid-related Telehealth flexibilities to continue and which to dispense with! To better represent our community’s telehealth needs, the Haystack Project has created a survey to learn specifically about our rare patients’ telehealth experiences these past two years.
Let’s not let Ultra Rare be left out of Congress’ deliberations on Telehealth!
Biomarkers and Cognition in Dup15q Syndrome
Children with 15q11-q13 duplications are at high risk for neurodevelopmental disabilities, particularly autism spectrum disorder (ASD) and intellectual disability (ID). Recently, dup15q syndrome became the focus of research in the UCLA Intellectual and Developmental Disabilities Research Center (IDDRC). The IDDRC is an innovative, multidisciplinary center for research on the core causes of developmental disabilities. IDDRC studies are designed to lead to possible interventions to treat causes as well as improve quality of life for patients and their families.
What is the purpose of the dup15q syndrome study?
This study aims to (1) quantify EEG biomarkers of dup15q syndrome and (2) characterize cognition and development in this population, using standardized behavioral testing, play-based assessments, eye tracking, and high density EEG. The overarching goal is to find EEG biomarkers and specific behavioral features that may serve as targets for behavioral and pharmacological intervention.
The study will involve approximately 1-2 days of testing, including parent questionnaires, behavioral evaluations of the child, and an awake, high density EEG. We will provide each family with detailed written and verbal feedback on the behavioral testing.
Whom do we want to recruit for this study?
Our goal for this study is to recruit children ages 1-18 with either interstitial or isodicentric 15q11-q13 duplications.
Whom do I contact?
Families who wish to learn more about this study, please contact:
Careese Stephens
UCLA IDDRC
UCLA Center for Autism Research and Treatment
cmstephens@mednet.ucla.edu
www.jestelab.org
Genetic Cell Repository
CHROMOSOME 15Q11.2-13.1 DUPLICATION CELL LINES
The National Institute of General Medical Sciences (NIGMS) Human Genetic Cell Repository, Coriell Institute for Medical Research
 In the study of human genetic disorders, samples from patients and their families are a critical resource for researchers, however it is often a major hurdle to identify families with specific chromosome abnormalities, like idic(15) and int dup(15). Thus many researchers rely on tissue and cell “banks” for access to samples from patients with a specific diagnosis. These samples are often in the form of a “cell line”, which are derived from a blood sample that has been specifically treated to allow the cells to be grown in culture for extended periods of time. Our lab has established cell lines for most of the samples that we have obtained for our study of chromosome 15q duplications. We would like to submit them to the NIGMS Human Genetic Cell Repository at the Coriell Institute for Medical Research to make them widely available to other researchers who are interested in working on chromosome 15.
In the study of human genetic disorders, samples from patients and their families are a critical resource for researchers, however it is often a major hurdle to identify families with specific chromosome abnormalities, like idic(15) and int dup(15). Thus many researchers rely on tissue and cell “banks” for access to samples from patients with a specific diagnosis. These samples are often in the form of a “cell line”, which are derived from a blood sample that has been specifically treated to allow the cells to be grown in culture for extended periods of time. Our lab has established cell lines for most of the samples that we have obtained for our study of chromosome 15q duplications. We would like to submit them to the NIGMS Human Genetic Cell Repository at the Coriell Institute for Medical Research to make them widely available to other researchers who are interested in working on chromosome 15.
The NIGMS Human Genetic Cell Repository at the Coriell Institute for Medical Research in Camden, New Jersey, holds the world’s largest collection of human cell lines for use in research, which includes over 10,000 cell lines representing more than 500 different genetic disorders. They maintain and distribute thousands of cell lines and DNA samples from individuals with diverse genetic disorders and make them available to researchers around the world for a nominal cost. Their collection is supported by grants from the National Institutes of Health and several private foundations with specific support by the National Institute of General Medical Sciences. To learn more about the cell repositories at Coriell, please see their website: ccr.coriell.org.
Coriell’s repository collects biological samples and relevant clinical information from donors. All personal identifying information is removed upon arrival to the repository. In the catalog, basic information is provided to researchers ordering cell lines, including gender, race, the age of the individual when the sample was obtained, and if other family members are in the repository. However, all of the information is anonymized, meaning that there is no way for the researcher to know who the sample came from.
Families who are participating in our study can contact the NIGMS Human Genetic Cell Repository genetic counselor, Tara Schmidlen, to authorize us to share your cells. Be sure to let her know if you are part of our study, because if we have samples that can be shared, we will send the cells to Coriell so that you do not need to have another sample of blood drawn. If families are not enrolled in our study, but would like to provide a sample for the cell repository, you can also participate in this opportunity to expand access to idic(15) and int dup(15) cell lines by contacting Tara. Families living outside of the United States are also welcome to donate a sample to the repository. All interested families will need to sign the Coriell consent forms in order to have their samples in the repository. These forms are being generated and will be available from Tara Schmidlen.
Tara J. Schmidlen, MS, CGC
Certified Genetic Counselor
NIGMS Human Genetic Cell Repository
Coriell Institute for Medical Research
403 Haddon Avenue
Camden, NJ 08103
tschmidl@coriell.org
P:856-757-4822
F:856-964-0254
Understanding social communication and motor skills
Drs. Inge-Marie Eigsti, Holly Fitch, and Stormy Chamberlain from the University of Connecticut are doing a research study of social communication and motor skills in autism and genetic disorders of Chromosome 15.
What’s involved?
- We are interested in the development of social communication and motor skills.
- You can participate online – no need to leave home.
- Parents will complete a 40-60 minute online survey
- Families will receive $30
Who can participate? Anyone ages 3 – 80 who:
- Has access to an online computer and webcam
- Has a caregiver who can provide information
- Has Angelman syndrome, dup15q syndrome, or autism spectrum disorder
Contact us for more information:
Elise Taverna at (860) 486-3085 or angelman@uconn.edu
Has Your Child Been Diagnosed with Cerebral Visual Impairment (CVI), Amblyopia, or Strabismus?
The Smith-Kettlewell Eye Research Institute is conducting a research study looking at vision and brain function in children. They are currently recruiting participants with CVI, from 0-3 years of age, 4-18 years of age and adults under 65 years old.
Questionnaire:
An hour of your time could help Dr. Chandna’s team develop better diagnostic and intervention tools for children with CVI, amblyopia and strabismus. Please call and schedule your interview.
Phone Interview Includes:
- 52 multiple choice questions
- about 1.5 hour in length
- $20 US/15£ UK per hour by money transfer service
- conducted remotely with a trained researcher
Email: Seelab@ski.org
SUDEP Registries & Tissue Donation
When an individual with dup15q syndrome passes away from SUDEP (Sudden Unexplained Death in Epilepsy), their experience can help researchers learn more about this poorly understood tragedy. The Alliance encourages families to reach out to either of these SUDEP registries, which collect patient information and, when possible, tissue samples for research into the mechanisms of SUDEP and potential prevention measures.
We understand that tissue donation occurs at a particularly emotional time for loved ones and that it is a personal and difficult decision that will not be right for everyone. By making this generous donation, you improve the chances of finding a cure and better treatment options for these diseases. Dup15q Alliance applauds the AAN’s new SUDEP guidelines, while joining other advocacy organizations in urging expanded surveillance, research, and education.
SUDEP Tissue Donation Program (STOP SUDEP)
Dr. Alica Goldman and her colleagues at The Department of Neurology at Baylor College of Medicine have established a seizure disorder tissue bank, the SUDEP Tissue Donation Program (STOP SUDEP). This project is funded by The National Institutes of Health (NIH)/The National Institute for Neurological Disorders and Strokes (NINDS). Investigators collect blood and/or tissue samples from individuals who have passed away as a result of a seizure disorder.
Learn more about this program here.
Tissue donation is a very special gift, but is one about which you may have more questions. We seek to answer some of those questions with this FAQ:
STOP SUDEP Frequently Asked Questions
To register for tissue donation, contact Dr. Alica Goldman
The North American SUDEP Registry (NASR)
The North American SUDEP Registry (NASR) seeks to help identify risk factors and mechanisms leading to sudden unexpected death in people with epilepsy (SUDEP). The registry collects DNA, brain tissue, and clinical data (e.g., medical records, EEG) for scientific studies about the causes of SUDEP. NASR collects information on cases of SUDEP regardless of how long ago the registrant may have passed away. Even though NASR may not be able to collect brain or DNA samples for older cases, tissue from older surgeries, MRIs, EEGs, and other clinical information have proven incredibly useful.
NASR is a highly collaborative organization with representation from many lay organizations (Epilepsy Foundation, CURE, Danny Did, Dravet EU, etc.) and more than 15 international academic institutions. NASR is the largest and most collaborative SUDEP registries with more than 3 full-time employees and a detailed methodology for SUDEP determination and adjudication by several epileptologists. NASR has enrolled more than 250 SUDEP cases and has brain and/or biospecimens from more than 120 SUDEP cases. A guiding principle of NASR is that clinical data and biospecimens will be shared with scientific colleagues.
The North American SUDEP Registry (sudepregistry.org) collaborates with Dup15q Alliance and ATP in collecting clinical data (e.g. video EEG, MRI, interview of caregivers) on patients with dup15q syndrome and sudden death. Dup15q Alliance professional advisory board member, Orrin Devinsky, MD serves as the Principal Investigator. Affiliation: NYU Langone & Saint Barnabas Medical Centers, FACES
NASR Frequently asked questions
NASR 1-855-432-8555
Autism BrainNet
 The Autism BrainNet is a campaign to understand and treat a whole spectrum of pervasive developmental disorders, such as those caused by chromosome 15q duplications.
The Autism BrainNet is a campaign to understand and treat a whole spectrum of pervasive developmental disorders, such as those caused by chromosome 15q duplications.
Many of us have signed the back of our driver’s license to give permission to donate our organs after we die. We have been made aware of the importance of doing this in order to help another person, and have taken action in advance so that something good can come from our death. We generally don’t think about this kind of opportunity in regard to our children affected by a chromosome 15q duplication. Now there is a program that provides families an opportunity to donate the brain and tissue of a loved one who has just died so that researchers can access a critical resource for understanding chromosome 15q duplications.
REGISTRATION
Registering to donate brain tissue does not mean that anyone anticipates your child will die an early death. It does mean, however, that you are prepared to act if such an unexpected and tragic event were to occur. It also includes you to receive a quarterly newsletter that updates you and your family on autism discoveries where brain tissue has a significant impact.
Brain tissue recovery is coordinated nationally by the Autism BrainNet, a network of 4 research sites across the United States. You can register at www.takesbrains.org/
In the event of a death, contact the 24-hour hotline number: 1-877-333-0999 for immediate assistance. You will be put in touch directly with the local site who will make the appropriate arrangements. You must initiate the call.
All inquiries are treated with absolute confidentiality.
Although many scientists have become aware of the importance of understanding what causes the symptoms of idic(15), a major roadblock to further understanding is the absence of brain tissue. Scientists are able to be creative and work with cells from blood and brain imaging, but access to studying the impact of idic(15) on the brains of affected individuals is vital.
— Dr. Edwin Cook Jr, Dup15q Alliance Professional Advisor
The Autism Tissue Program has a world-wide network of researchers seeking to understand how and why the brains of affected individuals are different. The brain is the locus of the behaviors associated with chromosome 15q duplications (repetitive and restricted activities, lack of communication and social skills) and the seizures experienced by over half of affected individuals. Brain tissue is vital because it is the only way to see individual brain cells and read the DNA and genetic messages to get a better understanding of chromosome 15q duplications.
Brain tissue will be tremendously valuable for understanding the ways that duplication of chromosome 15 affects brain development and function, which will hopefully help us provide better treatment options for patients with dup15q syndrome. It is difficult to think about the potential death of a child. However, thinking about it in advance allows the family to discuss the issues surrounding a brain tissue donation without the incredible emotional stress that occurs when a child has died. Advance planning for a tissue donation is critical.
— Dr. Carolyn Schanen, Dup15q Alliance Professional Advisor
Organ donation is a highly personal decision that has the power to accelerate research and transform the lives of countless families affected by chromosome 15q duplications. It reflects a family’s choice to benefit others and honors the complex and challenging life of the affected child. You can make a tremendous difference in research into chromosome 15q duplications by participating in this program.
A pathologist performs the procedure and coordinates with the funeral director. Your child is treated with the utmost respect and dignity. The procedures used to obtain brain tissue will not affect any funeral arrangements, including viewing, that you wish to make. The Autism BrainNet assumes all costs related to obtaining tissue.